How the Heck Does a Heat Pump Get Heat from Cold?!

It’s been cold out lately, so I’ve been talking about heat. When I talk to people about how heat pumps can keep your home warm in winter, they usually get a look of bewilderment on their faces. How is that even possible, they ask. No one ever tells me that they’re confused about how a refrigerator or air conditioner works, even though it’s the same exact process – moving heat from a cooler area to a warmer area.
The direction of heat flow
My guess is that we have blinders here because of our personal experience with cold outdoor temperatures. When we go outside in winter, our bodies have a much higher temperature, being at about 98.6°F, and we experience cooling. We personally always are cooled off by cold outdoor air, so it’s hard to imagine that that same air could ever heat anything else up.
Heat pumps really do get heat from cold air, though. So, to understand their operation, let’s start with the basics. I discussed heat flow, thermal energy, and temperature earlier this week, and in that article I said heat flows when you have a temperature difference (ΔT). So if you’re trying to get heat out of 40° F (5° C) air, what do you have to do? Put it in contact with something that’s at a temperature lower than 40°F! That’s the job of the refrigerant in a heat pump.
The science behind heat pumps
If you want a good, short description of the the science behind these devices, see my explanation of the refrigeration cycle. I’m going to focus on just one part of it here: the expansion valve (or metering device more generally).
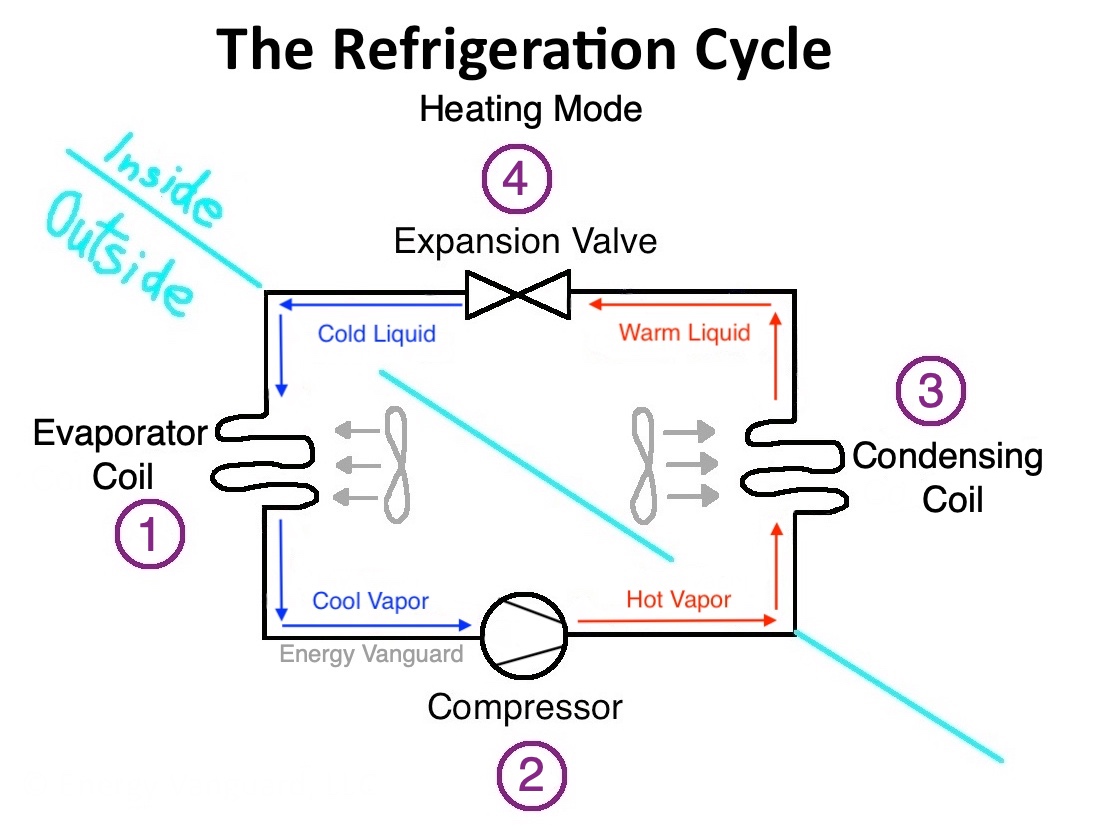
There are four basic processes in the refrigeration cycle. All are important, but in my opinion, the expansion valve is where the magic happens. Whether you’re using it for a refrigerator, air conditioner, or heat pump, achieving a low temperature is the key, and that’s what the expansion valve does for you.
You need colder than cold to get heat
 Here’s an example for you that you may have experience with, especially if you’re a serious bicyclist. CO2 cartridges contain carbon dioxide under high pressure. (Does this count as carbon sequestration?) When you use them to inflate a bicycle tube, for example, the cartridge gets very cold. Try it! It also works with aerosol cans like hairspray. This is a thermodynamic property of gases. When they’re allowed to expand freely, their temperature drops.
Here’s an example for you that you may have experience with, especially if you’re a serious bicyclist. CO2 cartridges contain carbon dioxide under high pressure. (Does this count as carbon sequestration?) When you use them to inflate a bicycle tube, for example, the cartridge gets very cold. Try it! It also works with aerosol cans like hairspray. This is a thermodynamic property of gases. When they’re allowed to expand freely, their temperature drops.
Same thing happens in a fridge, AC, or heat pump. The refrigerant is pushed through the expansion valve, and the temperature of the refrigerant drops—a lot. So, that cold outdoor air is actually the warmer stuff then, when it comes in contact with the outdoor coil of your heat pump. And yes, this can work even when the outdoor air is 5° F (-15° C).
And, as we know, heat likes to move from warmer objects to cooler objects. Once we get that heat from the air into the refrigerant, it’s just a matter of bringing it into the house and then transferring it into your home’s air.
So now the mystery is solved! It’s our old friend, the Second Law of Thermodynamics again.
Allison Bailes of Atlanta, Georgia, is a speaker, writer, building science consultant, and the founder of Energy Vanguard. He has a PhD in physics and writes the Energy Vanguard Blog. He is also writing a book on building science. You can follow him on Twitter at @EnergyVanguard.
Related Articles
The Magic of Cold, Part 1 – How Your Air Conditioner Works
How NOT to Use Your Heat Pump Thermostat
Can a Heat Pump Work in Minnesota?
NOTE: Comments are closed.
This Post Has 16 Comments
Comments are closed.

I have no idea what you just
I have no idea what you just said above, but you sure sound HOT saying it! And that would be the slang use of HOT! I think you said that if I’m cold, I should go outside and sit in my fridge. I’m game!
Actually, I just wanted to mention that we have a dachshund. And she is hiding under her blankie looking as pitiful as the one above at this very minute. I think I’ll go explain the Third Law of Thermodynamics to her – Stop Stealing the Blankies!
You’re a hoot, Alexandra! But
You’re a hoot, Alexandra! But why do you keep your fridge outside?
Thank you, once again,
Thank you, once again, Professor Bailes, for another stunningly clear explanation of something that confounds so many people. (I will leave y’alls definition of cold for another time. 40 degrees. Ha)
You’re welcome, Peter, and
You’re welcome, Peter, and thanks for the great compliment! It’s really hard to restrain myself from going deeper in my explanations. I could’ve explained the special properties of refrigerants that make them so useful, the latent heat involved in phase changes, and the other steps of the refrigeration cycle that I so casually dismissed. Believe it or not, this article could still use a little work to make it even clearer.
I figured someone from a colder area would say something about my choice of temperature, so it makes sense that it’s the guy from Maine. Actually, I know that 40°F isn’t cold, and we even had about three days this week when it stayed continuously below freezing here in Atlanta. That choice was made to avoid the discussion of balance points, supplemental heat, and the extra-special cold climate heat pump. Look for articles on those topics later!
Should my Trane heat pump
Should my Trane heat pump stop working if my house’s natural gas is disconnected? I thought it would still work but the temp inside has dropped to 55 degrees since my gas was disconnected. (Moral to this story…always open your mail, no matter how busy you are!! My gas was disconnected due to my forgetting to pay the bill online and my failure to open recent mail received from GNG warning me of impending disconnection…I paid the gas bill in full yesterday but the “first available” date for reconnection isn’t until next Thursday!!!)
Kimberly, if your heat pump
Kimberly, if your heat pump is connected to your furnace as part of a dual fuel system, you’ll probably get no heat. Since the heat pump and furnace cannot run at the same time, dual fuel systems have an outdoor thermostat that tells the system which one to use. When the temperature goes below the setpoint, the furnace operates. If the furnace can’t operate because of lack of gas, it won’t automatically switch to the heat pump. You’ll just get no heat.
If, on the other hand, your heat pump and furnace are two separate systems serving different parts of the house, the heat pump should still work, as it’s not connected to the furnace. It probably won’t keep your whole house warm, though, since it’s set up to serve only one zone.
It sounds like you may be out of luck till next Thursday. Got any friends or relatives who can take you in?
Thanks so much for your help!
Thanks so much for your help! I had the heat pumps installed several years ago when my HVAC units needed to be replaced, and as far as I know I don’t have a separate gas furnace, but I’m not getting any heat from either the upstairs or downstairs heat pump, so I must be wrong about that…must be tied into a gas system in some way. This’ll teach me to put off reading the mail! Thanks again for your reply. I just discovered this site and I think it’s a great resource!
Thanks for the excellent
Thanks for the excellent explanation. My question is “how cold will a heat pump still be effective?” I recently built a new house and had 2 13 SEER/7.8 HSPF units installed. They both have electric heat strips for backup. I’ve heard a lot of different numbers (temp) that a heat pump can operate at. Some claim that heat strips kick in automaticaly when temps get into the uppper 20s, some don’t kick in automatically at all. The other night temps got down around 11 degrees(f). The thermostat never indicated that heat strips came on (emerg. heat). System ran almost continuously but kept the house at selected temp of 67. At 11 degrees outside, was the system still operating in heat pump mode or was it most likely on backup? How low can a heat pump go?? I understand that the heat producing capacity diminishes at lower temps.
Thanks,
Bill
Good explanation of the basic
Good explanation of the basic thermodynamics behind heat pumps.
I’m in the process of an energy remodel and at the beginning we considered a ground source heat pump for our hydronic heating system, and a new air handling system for air conditioning. As we were planning also on having heat recovery ventilation (HRV), I thought we could get an integrated air handling system that would both ventilate and cool. Unfortunately, it is not to be found in the US (though they are available in Europe).
On top of this, the system required a large (80 gallon) buffer tank to hold water either chilled or heated to avoid having the pump switch on and off frequently. Our moderate climate in CA would not have the heat pump running all the time. The reason they need this is because the pump motors are not robust enough to handle modulating action, and tend to age more quickly if they are run in that mode.
Both of these, plus the high price, convinced me to stay with our gas fired hydronic boiler. It generates fossil carbon but I’ll just have to buy offsets for it (much as I dislike the idea of offsets).
If you want to know more about our energy remodel, check out the blog: http://netzerolife.blogspot.com.
Bill, you’ve got a lot of
Bill, you’ve got a lot of good questions, and I’ll be writing another article soon to address them. For now, though, let me give you a brief answer. Heat pumps can still pump heat down to at least the single digit temperatures Fahrenheit, possibly even below zero, because the boiling point of the refrigerant is down around -50°F.
As to when the supplemental heat kicks in, there’s something called the ‘balance point,’ the temperature at which the house needs exactly the same amount of heat that the heat pump can move into the house. When the temperature goes lower than that, you need supplemental heat. That temperature is usually in the mid-30s F but depends on the climate, the equipment, and the house. It’s hard to imagine that your equipment/house has a balance lower than 11°F, but if it does, then yes, the supplemental heat shouldn’t come on.
Look for more on this topic soon.
Kempf42, I don’t really know
Kempf42, I don’t really know anything about integrated heat pumps & HRVs, but it seems like I heard something not long ago similar to what you’re saying – that they’re a good idea in theory but don’t work so well in practice.
Thanks for the link to your blog. I checked it out, and it looks like you’ve got an interesting project going there. It also reminds me of when I was building nearly a decade ago and having to make all those decisions about products, materials, and methods. It takes a lot of work and research to do it right, and even then it’s still difficult. Keep up the good work!
We live in OH and just
We live in OH and just received our Dec/Jan all electric house bill.It shocked us, $507. Never that high in 5 years. Called my electric co. and was told to turn off my supplemental heat and just heat the house by heat pump. My furnace man told me to switch to supplemental when below freezing, so I had the supplemental switch on for all of December, Hence the high bill. Will it hurt my furnace or the heat pump to just run the heat pump without supplemental heat when the weather is below freezing? I can’t affort another bill like that! I do not want to damage this furnace, it’s about 40 years old. It’s a Trane. Seems to be warm enough in here, i keep it at 68 degrees. Need your imput, thanks, Ginny
Ginny, I think when you say
Ginny, I think when you say ‘supplemental,’ you mean ’emergency,’ right? Doesn’t the thermostat setting you had selected for December say ’emergency heat’? The only time you want to select that is when your heat pump is not working. In other words, if you have the thermostat set to ‘heat’ and the house doesn’t warm up at all, then you use ’emergency heat.’
Heat pumps cannot keep up with the heating load in most houses when the temperature gets below a certain point. They have built in supplemental heat to add to what the heat pump does. The supplemental heat is electric resistance heat, which is not very efficient.
If you’re using it only to supplement the heat pump, that’s not so bad, but when you set the thermostat to ’emergency heat,’ you’re bypassing the more efficient heat pump and using only the strip heat. That’s why your bill was so high in December.
My past electric bill was
My past electric bill was $211, which is for a small basement apartment in DC. I thought that seemed high until I realized I had a heat pump and that raising the heat from 60 to 68 each day is not economically wise.
I am now keeping my thermostat at 68 degrees during the day and night. My question is if it’s common for the heat pump to come up regularly during the day (handful of times each hour) to keep my apartment around 68 degrees? I am worried that it comes on too much.
Also, is it really that costly for me to turn up the heat only one or two degrees during the day if I need it warmer? I worry that even adjusting the thermostat slightly, will result in high heating bills.
Thanks,
Shaun
Ok, so all this talk of heat
Ok, so all this talk of heat pumps and yes we have one here at our 1978 Apt building…HOW do we go about telling management and or Corporate, that these things are old ..I mean OLD…the coils are all bent inside (the apt) we have ONE ceiling vent for the LIVING,DINING And Kitchen area..(which is placed above sliding single door) I have cleaned the inside coils with a soft brush and has helped some! BUT geesh louise, these run forever! I will set thermastat on 65 and it will run for 2 hrs? or more? IS this NORMAL?
Well stated information!
Well stated information!