Heat Rises…and Falls — Stack Effect, Air Movement, & Heat Flow

Heat rises. Everyone knows that, right? It’s absolutely true. Heat does rise. The problem is that sometimes people say this as if the flow of heat is driven by its wanting to rise. It’s not. Heat can move up, down, or sideways, depending on the situation. What the laws of thermodynamics tell us is that heat moves from areas of higher temperature to areas of lower temperature. Put a torch to the top of a steel pole, and heat will travel downward by conduction. So, temperature difference is really what drives heat to move in any given direction.
When you’re dealing with fluids, you have to account for density and buoyancy as well. Air is the fluid we live in, and this time of year we spend a lot of money pumping heat into it in our homes and workplaces. When we heat air, the molecules jiggle and zip around faster, which causes them to spread out. When a mass of air takes up more space, it has a lower density. When you have a lower density fluid immersed in a higher density fluid, the lower density fluid rises and the higher density fluid falls.
Think of air bubbles in water, as shown in the photo above. Think of a helium balloon. Think of a hot air balloon. Now, imagine an object with higher density immersed in a fluid. Put Wile E. Coyote’s anvil in the air above his head, and it turns him into a pancake.
The point here is that it’s easy to get confused by heat in the building science of air movement. Warm air rises when it’s surrounded by cold air because of its lower density. Yes, that’s due to heat, but density is the main factor causing the movement here. The name for this phenomenon is stack effect. Two factors affect how much stack effect a building experiences:
- Temperature difference between inside and out (because density depends on temperature)
- Height of the building
The problem with stack effect in buildings is that buildings aren’t vacuum chambers. They leak. Obviously, a house isn’t going to start floating up into the air like a balloon (although I recall with great fondness the Disney movies of my childhood that showed such magical events). But the low density air inside the house will move up and out into the cold, dense winter air when given the chance.
Try this experiment if you don’t believe me. Open your pull-down stairs or scuttle hole to the attic on a cold day when your home is warm. Climb up into the attic and then put your face over the hole. You’ll feel the stack effect pushing lots of warm air into the attic.
So, in winter, the warm, low density air inside your house wants to rise…if it can. If your house has no leaks, the warm air can’t escape and do its thing. There’s still a pressure difference across the building envelope, but that’s OK if the air barrier’s good. Positive pressure inside the house with nowhere to go because there are no pathways.
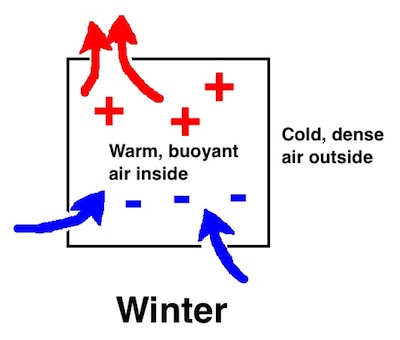
What happens in reality is that homes leak. Your nice warm air finds way to leak out (exfiltration) and cold air leaks in (infiltration). Because of its lower density, the warm air will leak out the top of the house if there are leaks there. When a cubic foot leaks out, however, it has to be made up by a cubic foot leaking in. As the warm air leaks out at the top, cold air leaks in at the bottom. The leakier your house is, the more temperature difference you’ll notice between the top and bottom of the house.
All this happens because the warm air inside your home in winter is less dense than the cold air outside. In summer, the dense air is inside your home because that’s where the temperature is lower, especially if you’re air conditioning your home. What that means is that leaks in your house bring warm air in at the top and allow cool air to fall out at the bottom.
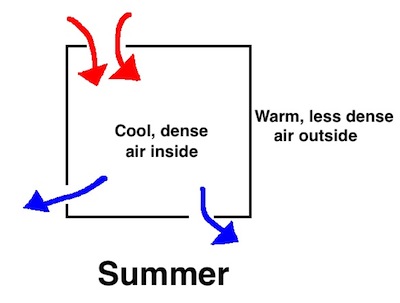
Ah, warm air falls! Heat sinks. That old expression, “Heat rises,” is not a basic truth after all. As with many aspects of building science, you have to look at the full context to understand what’s going on.
Followup Article
Who Knew the Stack Effect Could Be So Controversial?
Related Articles
What Is Pressure? – Understanding Air Leakage
Rats to You, Daniel Bernoulli! – Understanding Air Pressure (with a cool video!)
Infiltration Occurs at the Surface, Not in the Volume
It’s the Hole – Understanding What a Blower Door Is for
Photo of water bubbles by Christian Haugen from flickr.com, used under a Creative Commons license.
This Post Has 27 Comments
Comments are closed.

Terry Brennan and Bill Turner
Terry Brennan and Bill Turner (Camroden Associates and Turner Engineering) have a fantastic reference document written about 10 years ago on Stack Effect and Planned and Unplanned Airflow. Email me a request and I will send you the document.
Keep in mind that these same energy principles are the reasoning behind where the vapor barrier goes in an exterior assembly based on climate zone. The proclivity for which side of the assembly the highest concentration of heat and moisture most typically can be found dictates where the point of condensation in the assembly should be located.
Nicely written, clear
Nicely written, clear explanatory post. BTW, I used to scuba dive, and we’d spend much time practicing how to make those air bubble rings during boring decompressions 🙂
Matthew C.
Matthew C.: I don’t think I’ve seen that paper, but since Terry Brennan is one of the most knowledgeable people in the world when it comes to air movement, I’d certainly like to see it.
John P.: Thanks! So the ring bubble is a scuba thing, eh?
Alison, nicely put. To be
Alison, nicely put. To be picky I try to avoid saying that heat rises for other reasons. I say that heat moves toward cold. This is particularly obvious with conduction and convection. I do say that “Warmed Air” has a tendency to rise because it is lighter (convection). Your observation that in the warm months convection currents can draw warm air into the conditioned space from an attic is an important and often neglected point. I for one will be more careful with my use of the phrase “Warm Air Rises.” Thanks for the post.
Hi Alison,
Hi Alison,
Nice blog, but I would like to join Armand in being concerned about the picky details. The issue of warm air rising has created much confusion among energy professionals and home owners. In an effort to clear the air, so to speak, I would like to discuss better wording that may help to correct this long ignored science. Warm air moving up is indeed what we see, but warm air has no internal energy that makes it move up or down. It is entirely controlled by gravity. Much more to be said.
Bud
Armand M.:
Armand M.: I don’t have a problem with saying that heat rises because I usually follow that up with “and heat falls, too.” What are your other reasons for avoiding it?
Bud P.: Yes, we can quibble about details and semantics on this topic for a long time. If warm air moves upward, we can say that it’s rising. Because it’s warmer than the surroundings in winter and leaves behind cooler, denser air at the bottom of the house, we can say heat rises. I don’t have a problem with that. Buoyancy is the relevant principle here, and Archimedes figured it out thousands of years ago. What is the better wording that you suggest?
Thanks for the prompt reply,
Thanks for the prompt reply,
One in particular is when the statement “warm air rises” is followed with “it then pulls in its replacement air”. Since warm cannot rise of its own accord, there is no pulling effect behind it and thus the implied negative pressure. Attic ventilation is an example where you often hear that the warm air rises to exit the upper vents and pulls in its replacement air.
Wording for our new energy correct language is just being put together, but one initial thought is to avoid the “pull” word and use “displacement air” to replace “replacement air” when describing the inflow of colder air.
The objective here is to dispel the notion that warm air is leading the way in the process of convection, when in fact it is the cold air that initiates the process.
It seems like semantics until you try to calculate the moving forces behind stack effect and find there is no equation for the mythical upward force from the warm air. Buoyancy is indeed the principle involved and it is the difference in the weigh of warm vs cold air.
You did use the term “push” when describing the airflow into the attic, which is correct.
Bud
Bud, I like your words…
Bud, I like your words…”it is the cold air that initiates the process”
it reminds me of Julius Sumner Miller:
“the less dense air is pushed up by the colder air”
http://www.youtube.com/watch?v=S57nIs503fA&feature;=results_video&playnext;=1&list;=PL4795B8F0927BE8B9
Allison, well put. I always
Allison, well put. I always make the point that they are not “heat balloons.” They’re “hot air balloons” and try to apply my mentor’s adage that in summertime (in some climates) you have to turn your building science upside down and inside out. Also, I have a link to an excellent photo of a house-shaped hot air balloon. E-mail me a request if you’re interested.
Allison, how can I make the
Allison, how can I make the links that I enter “hot”
Bud: I’m
Bud: I’m not sure why you think that “cold air that initiates the process” when it’s really pressure differences that move air across the building envelope. At the top of the house, there’s a pressure difference across the ceiling, and air moves from the area of high pressure (inside the house) to the area of low pressure (the attic). At the bottom, the pressure difference is reversed (positive outside, negative inside). Leaks at both the top and bottom are necessary for air to keep moving. If the top is airtight, the pressure difference at the bottom goes to zero. If the bottom is airtight, the pressure difference at the top goes to zero.
Regarding the word ‘pull,’ I don’t see a problem with it. If air leaks out through the top of the house, more warm air rises inside the house to take its place. That makes the pressure at the bottom of the house more negative, thus ‘pulling’ in more air from outside.
Great article Allison. I will
Great article Allison. I will share it, so some of my green building buddies can read it.
Funny thing about the semantics conversation is that usually when this is brought up, it leaves out the comprehension level of the average home owner. Which is whom I serve. I already see the blank stares on their face when I try to explain air infiltration based on pressure planes and differentials and the stack effect. If I didn’t know what you were talking about, I would have had a hard time understanding the article, let alone the following conversations.
I disagree. I just finished
I disagree. I just finished reading Dr. Bailes’ article. I’m not the most technical person and I know next to nothing about building science. But I found the article clear and easy to understand. I’m a visual person and the drawings helped me to grasp a concept I’ve never even thought about. I don’t think even my husband (David) could have done a better job explaining this to a lay person.
Allison, you said: I
Allison, you said: I’m not sure why you think that “cold air that initiates the process”
When you insert a mass of cold air into the middle of a warm room, the stack of air passing down through the cold air will have a greater pressure at the bottom than an adjacent stack of air passing through the warm room. The pressures at the top will be identical because each will have an identical amount of air above them. As the cold air moves due to the difference in pressure at the bottom it creates a reduced pressure at the top resulting in the warm air being pulled in the replace it.
It is obvious that both must move, but the warm air cannot rise by itself and therefore cannot be credited with pulling the cold air in behind it. Convection is also the movement of air and since it is the increased pressure that determines the direction of that movement, I give credit to the cold air.
You said, and this is an example of the conventional thinking we must change:
“If air leaks out through the top of the house, more warm air rises inside the house to take its place. That makes the pressure at the bottom of the house more negative, thus ‘pulling’ in more air from outside.”
This is not correct. The negative pressure we see at the bottom of our homes is due to the reduced pressure from a stack of air passing down through the house that now includes the warm air inside. Since the colder air outside our homes is heavier, it is applying a positive pressure on the bottom of our homes which makes the inside look negative. That difference in pressure then forces cold air into our homes pushing the warm air up and forcing it out the top with a positive inside pressure. Here is my test page where I’m trying to create a working explanation of stack effect which walks through the creation of these positive and negative pressures.
http://myenergyworkshop.homestead.com/hot-air.html
Bud
Bud: You
Bud: You said: “…this is an example of the conventional thinking we must change.” I’m still waiting to be convinced that there’s reason or need to change.
The change is that we need to
The change is that we need to recognize that warm air does not rise and “pull” in its replacement air. A good example of a misconception that has evolved from this pulling concept is the short-circuit theory when gable vents are left in place along with soffit and ridge vents. As the theory goes, the exiting warm air will pull its replacement air from the gables instead of from the soffits where we want it. Thus the soffit air flow will have been short circuited. The reality is, the air that flows in the soffits is going to happen regardless of where it flows out. It is not a pulling effect that is creating the pressure across the soffits but the barometric differences between inside and out. The link provided does get a little more involved as I am putting numbers on the stack effect pressures, but it does illustrate that the negative pressures we see in our basements are the result of barometric differences and not a vacuum created by exiting air.
Since this is getting long, you are welcome to use my email and we can continue this over a longer period of time. This energy correctness is just getting started and will require time to find changes that will be comfortable to all. Your contributions would be greatly appreciated.
Bud
Bud, Thanks for posting your
Bud, Thanks for posting your unconventional thoughts…I like what you are saying…and I like the “test page” that you posted
Thanks for the video, he is
Thanks for the video, he is very entertaining and does make a definite statement about cold air is the force behind warm air moving up.
Bud
The comment by Jon LaMonte
The comment by Jon LaMonte brushes closest to what should be the point: demonstrating to clients a basic understanding and importance to controlling the air flow in their buildings and homes. Technical precision of the processes occuring is not needed hear and would interfere with the intended COMMUNICATION. I(the client) don’t care what came first, the chicken or the egg. I need the eggs to make french toast.
Clarence: Thanks for the
Clarence: Thanks for the comment. I love reading people’s blogs on these types of subjects. Often the writer forgets who the ultimate entity is that is being served, the home or building owner. That is why I appreciate Allison’s blogs so much because they not only educate and create a useful forum for us energy nerds, but they are also usually clear enough for the lay person to understand and learn from as well.
I always get a kick out of the comment sections because it is often obvious that that ultimate purpose is lost in translation or semantics.
Nice job on keeping it simple
Nice job on keeping it simple Allison!
Bud & others are
Bud & others are welcome to comment here:
What must we say?
We could call this thread
We could call this thread positve pressure.dp
Allison, You always seem to
Allison, You always seem to choose your words very carefully.
I am curious if Bud Poll’s November blog at Home Energy Pros prompted or influenced the wording in this blog?
John B.: I
John B.: I did see Bud’s discussion on HEP and wasn’t swayed by his argument. I’ve got another article on this topic coming out tomorrow morning, so check back then.
If I said “Cars are blue
If I said “Cars are blue…” I would certainly not be ‘wrong’ either, but I would also not be helpful. If I qualified that by further stating “… and cars are other colors too.” would perhaps be even less helpful. It would however, in the world of BS, provoke comments and drive blog traffic and prompt some individuals who want to be helpful to make comments where they would otherwise not. Mission accompished Allison. 😉
I try to never say “heat rises” but instead here’s how I choose to handle it.
I say “We know there are three forms of heat – convective, conductive and radiant – and they are all working together. When warm air rises, that is primarily concerning the convective form. We’re first aiming to get a handle on that one because it can dominate heat loss if not effectively controlled.”
Its helpful, I find, to quickly and up front take the time with a client to explain the 3 forms. I find that most people ‘get it’ and it provides foundation for a host of other explanations that eventually follow.
If I said “Heat rises”, that is the only part anyone would remember (no matter what qualification you append to it). The proof is likely that in your own mind, as in mine, it gained a strong foothold in my earliest childhood memories (long before I knew how much BS I was in for).
Please know that I have long ago curtailed my desire to police so called improper use of words like “concrete vs. cement” or “irrespective vs irregardless” or “fuel vs gas” because they don’t really affect the understanding of a concept.
On the otherhand, when it comes to BS, clarity and understanding has along way to go and needs support by eliminating that unhelpful phrase.
Long live “Warm air rises.”
Nice blog. Since it serves to clarfy more than confuse the end sum is always positive. 🙂
Lol… seems like the point
Lol… seems like the point is “one leaves and the other comes in at the same time.” Is there a balance, always? If it is cold in a building and hot outside, that cold air will join the heat. And the opposite. Where is the heat if an enclosure is 32f and so is the outside?